- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2016
- Matter at extremes
- Phosphorus becomes five-coordinated at high pressure
Phosphorus becomes five-coordinated at high pressure
The behaviour of the spin-Peierls compound titanium orthophosphate, TiPO4, was investigated up to a pressure of 56 GPa using single-crystal X-ray diffraction. Four high-pressure phase transitions lead to the phase containing phosphorus coordinated by five oxygen atoms.
Phosphorus plays a key role in the chemistry of life and many technological applications. Unusually high coordination numbers of cations like phosphorus are often associated with interesting physical properties such as high density, ultra-high hardness or low compressibility, which are important for engineering applications. So far, examples of a phosphorus atom coordinated by five oxygen atoms are known for liquids and molecular compounds, but were never found in inorganic network compounds, which usually contain stable PO4 groups.
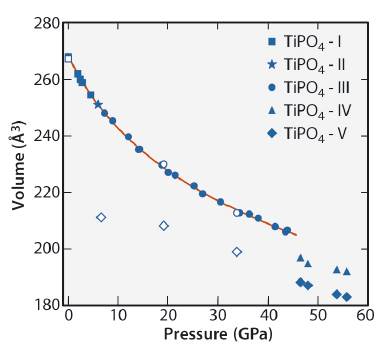
In this study, we investigated the high-pressure behaviour of titanium orthophosphate (TiPO4) by means of single-crystal X-ray diffraction at the beamlines ID09A of ESRF (now ID15B) and P02.2 (DESY, Petra III, Hamburg). TiPO4 belongs to the family of phosphates MPO4 (M = Ti, Tl, V, In) that crystallise in the CrVO4 structure type. TiPO4 is a very peculiar member of the CrVO4 structure family, because of its low-dimensional magnetic properties. On cooling, it undergoes normal–incommensurate–commensurate phase transitions towards a so-called spin-Peierls state. [1] The spin-Peierls transition is a magneto-elastic transition that occurs in quasi one-dimensional antiferromagnetic materials leading to the formation of spin singlets and dimerisation of the chains of antiferromagnetic atoms. Application of pressure to the spin-Peierls systems may lead to various magnetostructural transitions replacing the phase transitions predicted by classical crystal chemistry considerations. Indeed, we have found that the moderate compression of TiPO4 leads to the formation of the incommensurate phase TiPO4-II, which undergoes the lock-in transition towards commensurate TiPO4-III above 7 GPa. Both TiPO4-II and TiPO4-III contain Ti-Ti singlet dimers inherent to the spin-Peierls state [2,3]. This result improves our understanding of the spin-Peierls transitions and reveals new possibilities to examine this transition without low temperatures.
 |
|
Fig. 127: Pressure dependence of the unit cell volume. For TiPO4-II and for TiPO4-III, the volumes given are the volumes of the average structures. Open symbols correspond to the decompression experiment. The solid curve represents the fit of the third-order Birch-Murnaghan equation of state to the experimental data. |
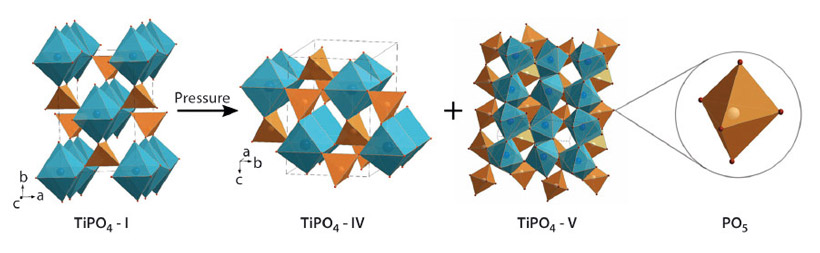
Compression above 46 GPa leads to the transformation to the coexisting phases TiPO4-IV and TiPO4-V with the volume drops of 4.2 and 8.0% respectively (Figures 127, 128). The structure of TiPO4-IV consists of TiO7 trigonal prisms capped on a single rectangular face. They share their trigonal faces, thus forming infinite chains along one of the crystallographic directions while Ti atoms along the chains remain dimerised. The chains are interconnected by PO4 groups via common edges and vertices.
|
Fig. 128: Crystal structures of TiPO4-I, TiPO4-IV and TiPO4-V. TiO6, TiO7 and TiO8 polyhedra are blue, PO4 and PO5 polyhedra are orange. |
The structure of TiPO4-V is composed of PO5 trigonal-bipyramids which form chains along the [1 0 1] direction through shared apical oxygen atoms. The chains are interconnected with each other by Ti atoms, linked to the equatorial oxygen atoms of the PO5 groups, thus forming layers parallel to (1 0 -1), such that the Ti atoms are confined between these layers. Ti atoms have highly distorted snub disphenoid coordination.
The experimental demonstration that phosphorus could form PO5 polyhedra suggests that similar compounds may appear as intermediate (metastable, short- lived, and hardly or not at all detectable) products of different chemical processes involving phosphorus and oxygen. Experimental evidence of phosphorous penta-coordinated by oxygen may open new routes in high-pressure phosphorous chemistry.
Principal publication and authors
High-pressure phase transformations in TiPO4: a route to pentacoordinated phosphorus, M. Bykov (a, b), E. Bykova (a), M. Hanfland (c), H.-P. Liermann (d), R.K. Kremer (e), R. Glaum (f), L. Dubrovinsky (a) and S. van Smaalen (b), Angewandte Chemie Int. Ed. 55, 15053-15057 (2016); doi: 10.1002/anie.201608530.
(a) Bayerisches Geoinstitut, Universität Bayreuth (Germany)
(b) Laboratory of Crystallography, Universität Bayreuth (Germany)
(c) ESRF, Grenoble (France)
(d) Deutsches Elektronen-Synchrotron (DESY), Hamburg (Germany)
(e) Max Planck Institute for Solid State Research, Stuttgart, (Germany)
(f) Institute of Inorganic Chemistry, University of Bonn (Germany)
References
[1] A.A. Buzdin et al. Soviet Physics Uspekhi 23, 409 (1980).
[2] J.M. Law et al. Phys. Rev. B 83, 180414(R) (2011).
[3] M. Bykov et al. Phys. Rev. B 88, 184420 (2013).



