- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2016
- Structure of materials
- The nature of electrocatalytically active sites elucidated by X-ray diffraction
The nature of electrocatalytically active sites elucidated by X-ray diffraction
Catalysts based on porous hollow PtNi/C nanoparticles were prepared through a one pot synthesis followed by annealing. X-ray diffraction and electrochemical measurements revealed that the structurally disordered areas of the PtNi shell are highly active for the oxygen reduction reaction, a key reaction for proton-exchange membrane fuel cells.
In times of ongoing depletion of fossil fuel resources, it is more important than ever to find alternative energy sources. Low-temperature proton-exchange membrane fuel cells (PEMFCs) are electrochemical devices that convert the chemical energy of a reaction into electricity. They are the most promising candidates to generate electricity for both mobile and stationary applications. However, before a switch of our society to H2-powered PEMFCs (H2 is the fuel used at the anode) can occur there are still many fundamental limitations that need to be solved such as the sluggish oxygen reduction reaction kinetics (ORR, the cathodic reaction) and the limited durability of the cathode catalyst in real devices. The best way to improve the ORR activity and decrease the catalyst cost currently appears to be with the use of carbon supported PtM nanoalloys (PtM/C, where M is an early or late transition metal) or core-shell nanoparticles composed of a Pt-enriched shell and a metallic or alloyed core. However, these catalysts are still far from the target 8-fold mass activity (A g-1Pt) improvement over state-of-the-art Pt/C required for PEMFC automotive applications.
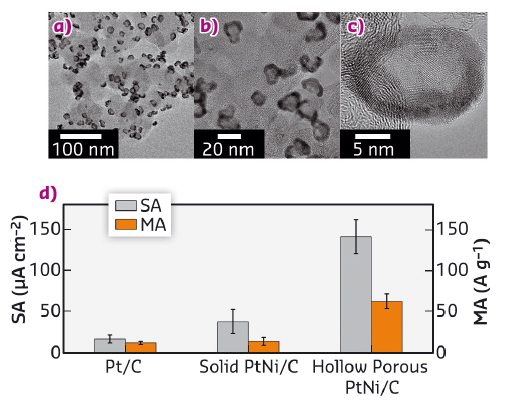
We have developed a simple one-pot synthesis of porous hollow PtM/C nanoparticles [1]. These nanoparticles are composed of a PtM shell surrounding a central void, which minimises the quantity of Pt contained in the catalyst. Moreover, their porosity allows oxygen molecules to reach the inner surface. The hollow PtNi/C nanoparticles (Pt:Ni 85:15 at.%) achieved 6-fold and 9-fold enhancement in mass and specific activity for the ORR, respectively over standard solid Pt/C crystallites of the same size. The catalytic enhancement was 4-fold and 3-fold in mass and specific activity, respectively over solid PtNi/C nanocrystallites with similar chemical composition, Pt lattice contraction and crystallite size (Figure 50). These results show that, beyond the classical strain and ligand effects, structural effects play a role in ORR electrocatalysis.
 |
|
Fig. 50: a), b), c) Conventional and high-resolution TEM images of hollow PtNi/C nanoparticles and d) specific and mass activity for the ORR measured at E = 0.95 V vs. the reversible hydrogen electrode. The specific activity and the mass activity are normalised to a cm2 of Pt or to a g of Pt, respectively. Potential sweep rate v = 0.005 V s-1, ω = 1600 revolutions per minute, T = 298 ± 1 K, 0.1 M HClO4. |
By combining high-resolution transmission electron microscopy (HR-TEM) and X-ray diffraction at beamline ID31, we have unravelled the crucial role of structural defects in the ORR activity. Conventional and high-resolution transmission electron microscopy (TEM) images of hollow PtNi/C nanoparticles displayed in Figure 50 showed that the PtNi shell surrounding the central void contains a high concentration of point defects (vacancies), planar defects (dislocations and grain boundaries) and bulk defects (voids, pores).
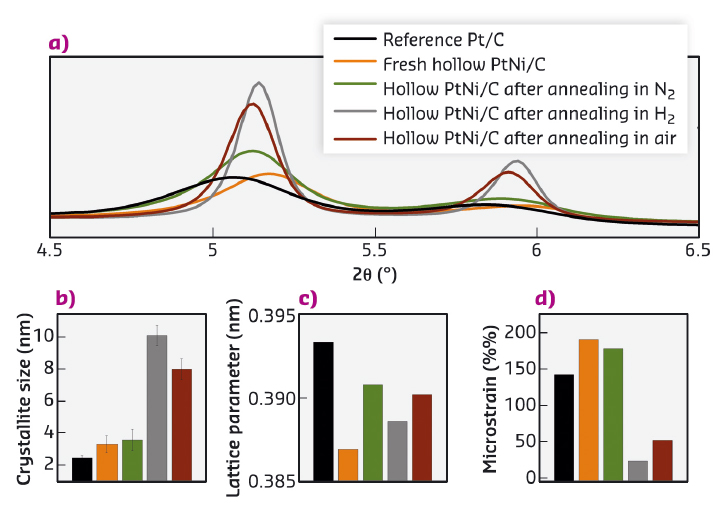
This highly defective nanostructure was confirmed by the high microstrain value (deviation of the inter-planar distance from its average value) derived from Rietveld analysis of high-energy X-ray diffraction (HR-XRD) patterns. Structural parameters of thermally-annealed hollow PtNi/C nanoparticles, which feature identical chemical composition to the mother catalyst but different morphology, crystallite size and lattice strain are displayed in Figure 51. Despite their relaxed lattice parameter, and their smaller crystallite size (parameters known to negatively influence the ORR activity), the materials featuring the higher microstrain values (that is the higher structural disorder) proved to be the most active for the ORR.
 |
|
Fig. 51: Structural parameters of fresh and thermally-annealed hollow PtNi/C electrocatalysts and of a commercial reference Pt/C. a) Synchrotron XRD patterns after linear heating from room temperature to 400°C at a rate of 2°C min-1, b) average crystallite size, c) lattice parameter and d) microstrain estimated by Rietveld analysis of the HR-XRD patterns. |
To gain further insight into the reactivity of these nanomaterials, in situ (i.e. under potential control) wide angle and small-angle X-ray scattering measurements during accelerated stress testing are planned. Combined with X-ray energy dispersive spectroscopy analyses these experiments should provide a detailed picture of structure-activity-stability relationships for this novel class of materials and should ultimately aid in the rational design of electrocatalysts with enhanced ORR activity.
Principal publication and authors
Defects do catalysis: CO monolayer oxidation and oxygen reduction reaction on hollow PtNi/C nanoparticles, L. Dubau (a,b), J. Nelayah (c), S. Moldovan (d), O. Ersen (d), P. Bordet (e,f), J. Drnec (g), T. Asset (b,c), R. Chattot (a,b) and F. Maillard (a,b), ACS Catal. 6, 4673–4684 (2016); doi: 10.1021/acscatal.6b01106.
(a) Université Grenoble Alpes, LEPMI, Grenoble (France)
(b) CNRS, LEPMI, Grenoble (France)
(c) Laboratoire Matériaux et Phénomènes Quantiques (MPQ), UMR 7162, CNRS & Université Paris-Diderot (France)
(d) Institut de Physique et Chimie des Matériaux de Strasbourg (IPCMS), UMR 7504, CNRS-Université de Strasbourg (France)
(e) Université Grenoble Alpes, Institut Néel, Grenoble (France)
(f) CNRS, Institut Néel, Grenoble (France)
(g) ESRF
References
[1] L. Dubau et al., ACS Catal. 5, 5333-5341 (2015).



