- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2017
- X-ray nanoprobe
- The complex crystalline structure of an oyster shell
The complex crystalline structure of an oyster shell
Biomineralisation processes produce outstandingly complex mineralised structures in living organisms. New structural features of a paradigmatic calcareous biomineral have been revealed by 3D Bragg ptychography, an X-ray microscopy method. This provides a better understanding of biomineralisation and supports recent biomineralisation models.
In many living organisms, biomineralisation processes regulate the production of the mineralised tissues such as bones, teeth and shells. Deciphering these mechanisms is of crucial importance for materials science, as it will provide bio-inspired strategies for the synthesis of nanostructured inorganic materials using soft chemistry and environmentally friendly processes. Such understanding is also important for paleoclimatology where biomineral proxies are used to perform paleoclimate reconstructions. The calcium carbonate biomineral is one of the most striking examples because theories arising from classical crystallisation (involving monomer-by-monomer addition) cannot explain the production of highly regulated calcareous crystalline biomineral structures such as those observed in sea urchin or pearl oyster. The production of this major constituent of the Earth’s crust is still poorly understood.
The present study was motivated by an apparent contradiction observed in biomineral structures: while calcareous crystallising species present a remarkable architectural diversity at the macro- and microscales, their sub-micrometric scale is characterised by a granular, but crystalline, structure [1]. Hence, a proper description of the crystalline features at this mesoscale level, i.e., over a few sub-micrometric (50-500 nm) granules, is a key to building realistic scenarios of biomineralisation. However, none of the currently used experimental approaches (electron- or X-ray-based diffraction microscopies) is able to provide access to the detailed 3D crystalline granule arrangement.
In 2011, a new approach, named Bragg ptychography, was proposed to image the crystalline properties of complex materials in 3D [2]. This microscopy method exploits the partially redundant information obtained by scanning a finite-size beam spot transversally to the sample, while measuring the corresponding 3D far-field intensity coherent diffraction pattern in the vicinity of a Bragg reflection. The 3D structure of the crystalline sample is further retrieved by inversion algorithms, which deliver a highly resolved image beyond the limit imposed by the size of the beam spot.
 |
|
Fig. 70: a) The pearl oyster shell (Pinctada margaritifera) and the investigated region (white rectangle). b) Enlarged view of the shell border showing the microscopic structure of the shell constituted of calcite prisms. c) X-ray Bragg diffraction pattern acquired with the ID13 X-ray nanobeam and integrated over the full prism area. The single-crystalline behaviour of the shell produces well-defined Bragg diffraction spots. |
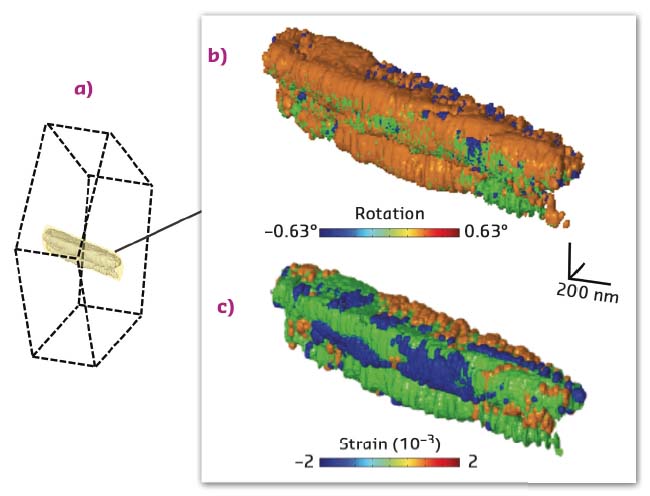
This cutting-edge synchrotron-based X-ray microscopy method was implemented at beamline ID13 [3] and used to reveal the details of the mesocrystalline organisation in calcite prisms, the generic mineral units of the pearl oyster shell (Figure 70). While these prisms are usually described as single crystals, the 3D image proves the existence of large iso-oriented and iso-strained crystalline domains, slightly different one from the other (Figure 71). These original results call for specific non-classical crystallisation pathways: the highlighted mesocrystalline properties support recent biomineralisation models, involving the partial fusion of oriented attached primary crystalline nanoparticles [4] or the formation of micrometric domains arising from liquid droplet precursors, successively transformed into an amorphous and then crystalline material [5].
 |
|
Fig. 71: Results of the 3D Bragg ptychography microscopy reconstruction. a) The retrieved volume (in yellow-grey) represents a small portion of the prism volume. b) 3D rotation and c) strain maps, showing the existence of crystalline domains within the single-crystalline-like biomineral. |
With the unique properties of the forthcoming upgraded ESRF source combined to the new (and fast) Bragg ptychography microscopy [6], the fine details of the biomineralisation processes will be soon within reach.
Principal publication and authors
Revealing crystalline domains in a mollusc shell “single-crystalline” prism, F. Mastropietro (a), P. Godard (a), M. Burghammer (b), C. Chevallard (c), J. Daillant (d), J. Duboisset (a), M. Allain (a), P. Guenoun (c), J. Nouet (e) and V. Chamard (a), Nature Materials 16, 946-952 (2017); doi: 10.1038/NMAT4937.
(a) Aix-Marseille Univ, CNRS, Centrale Marseille, Institut Fresnel, Marseille (France)
(b) ESRF, Grenoble (France)
(c) NIMBE, CEA, CNRS, Université Paris-Saclay, Gif-sur-Yvette (France)
(d) Synchrotron SOLEIL, Gif-sur-Yvette (France)
(e) GEOPS, Univ. Paris-Sud, CNRS, Université Paris-Saclay, Orsay (France)
References
[1] Y. Dauphin, J. Biol. Chem. 278, 15138 (2003); Y. Dauphin, Mineral. Mag. 71, 247 (2008).
[2] P. Godard et al., Nature Communications 2, 568 (2011).
[3] V. Chamard et al., Scientific Reports 5 9827 (2015); A. Pateras et al., Physical Review B 92, 205306 (2015).
[4] A.-W. Xu et al., J. Mater. Chem. 17, 415 (2007).
[5] M. A. Bewernitz et al., Faraday Discuss. 159, 291 (2012).
[6] S.O. Hruszkewycz et al., Nature Materials 16, 244 (2017).



