- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- X-ray Imaging
- Direct three-dimensional imaging of polymer–water interfaces by nanoscale hard X-ray phase tomography
Direct three-dimensional imaging of polymer–water interfaces by nanoscale hard X-ray phase tomography
Surfaces with special wettability can exhibit unique properties like self-cleaning or cell repellency and therefore attract much research interest in the quest to improve coating technology. Superhydrophobic and superoleophobic surfaces, in particular, have been studied intensively in recent years due to their extraordinary liquid repellent properties. Slippery liquid-infused porous surfaces (SLIPS) based on lubricant-infused porous polymer substrates [1] were found to have particularly good anti-bacterial and marine anti-biofouling properties. Visualisation of such surfaces and their interfaces to water or aqueous solutions have been attempted via confocal laser-scanning and via cryo-FIB/SEM microscopy but artefacts due to the required staining or freezing processes cannot be excluded.
To better understand such surfaces in their natural state, we investigated their structure on the micro- and nanoscale by three-dimensional (3D) hard X-ray phase tomography. To have a representative interface area, several mm thick samples were needed and therefore hard X-rays were used. Since water, lubricant (oil) and polymers provide weak absorption contrast, we used holotomography at the nano-imaging endstation ID22NI (now at ID16A) for propagation-based phase imaging of such interfaces. Multilayer-coated Kirkpatrick–Baez (KB) optics provided a tightly focused spot (50×50 nm2 H×V, FWHM) of monochromatic X-rays (E = 29.6 keV, ΔE/E ≈10-2). The sample was placed downstream of the focus in the slightly diverging beam, and, by varying the source-specimen and specimen-detector distances, the geometrical magnification M and thus the spatial resolution of the imaging setup could be adjusted. Fresnel diffraction patterns acquired with different M and effective propagation distances D in this so-called projection microscopy set-up allow retrieval of the phase shift introduced by the specimen with a high sensitivity that allows us to distinguish between water, polymer and oil after tomographic reconstruction of the specimen’s electron density.
As the material for our study, we chose porous poly(butyl methacrylate-co-ethylene dimethacrylate) (BMA-EDMA) surfaces, as described in [2]. The substrates of 5×25 mm2 surface area were imaged inside a polypropylene tube of 8 mm diameter that was filled with water. Each scan at all four propagation distances took around 45 minutes.
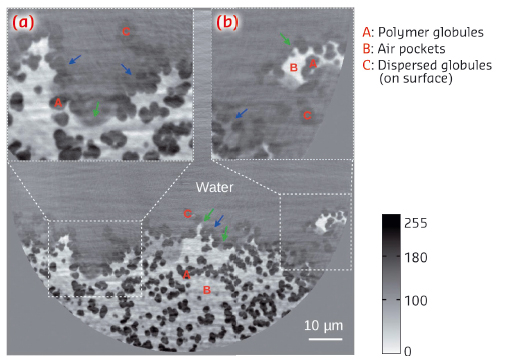
The 2D distribution of the electron density on a tomographic cross-section through the substrate is shown in Figure 68. Water, air and polymer globules of the porous structure can be distinguished. The visualisation of the structure (see also Figure 69) allows us to identify regions which coexist and follow two existing models that explain the hydrophobicity of a porous surface, namely the Wenzel state [3], where the increased surface area of the rough interface is responsible for its hydrophobicity or the Cassie–Baxter state [4] where gas trapped between the rough surface and water favours hydrophobicity.
 |
|
Fig. 68: Cross-section of the microporous BMA-EDMA surface on a glass substrate at an effective pixel size of 50 nm. Insets (a) and (b) are enlargements of the white dashed selections. Red colour labels A, B and C denote polymer globules, air pockets and dispersed globules, respectively. Air was trapped by the structured BMA-EDMA polymer. The coexistence of the Wenzel state (blue arrows) and Cassie–Baxter state (green arrows) was observed. A volume of 0.1×0.05×0.01 mm3 was rendered in 3D via grey level segmentation, see Figure 69a. |
 |
|
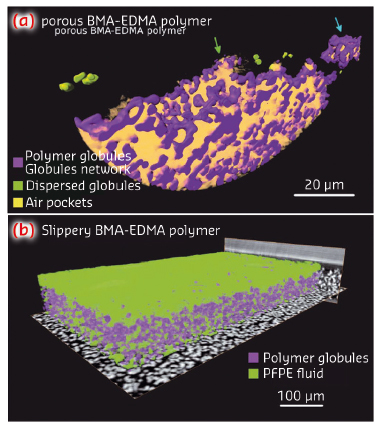
Fig. 69: a) Volume rendering of the local mass density of the porous BMA-EDMA surface, voxel size 50 nm. Blue and green arrows indicate Wenzel and Cassie–Baxter states, respectively. b) Rendering of the slippery BMA-EDMA surface with a voxel size of 0.32 µm. |
Figure 69 shows 3D visualisations of such interfaces at two different spatial resolutions in the nm-scale (a) and on the (sub-)µm scale. For the oil-infused surface in (b), such 3D data sets can be used to determine features such as the infusion quality or the spatial fluctuation of the thickness of the lubricant – here perfluoropolyalkylether (PFPE) oil – e.g., due to the anisotropic pressure imposed by the surrounding water environment.
Summarising, we have demonstrated non-invasive direct 3D imaging of surface–water interfaces on hydrophobic porous BMA-EDMA and slippery BMA-EDMA surfaces with hard X-ray phase-contrast nanotomography. The method provides isotropic volumetric structural characterisation of complex interfaces as well as bulk materials from sub-µm to nm scales whilst preserving the ambient conditions and representative specimen sizes. These advantages are of central importance to help understand the correlations between surface properties and their physical micro- or nanostructures which are needed to identify present limitations of such coatings.
Principal publication and authors
Y. Cheng (a), H. Suhonen (b,c), L. Helfen (a,b), J. Li (a,d), F. Xu (a,b), M. Grunze (a,d), P.A. Levkin (a,d) and T. Baumbach (a), Soft Matter 10, 2982-2990 (2014).
(a) Karlsruhe Institute of Technology (Germany)
(b) ESRF
(c) Presently at: University of Helsinki (Finland)
(d) Department of Applied Physical Chemistry, University of Heidelberg (Germany)
References
[1] T.-S. Wong, S.H. Kang, S.K.Y. Tang, E.J. Smythe, B.D. Hatton, A. Grinthal and J. Aizenberg, Nature 477, 443–447 (2011).
[2] P.A. Levkin, F. Svec, J.M.J. Fréchet, Adv. Funct. Mater. 19, 1993-1998 (2009).
[3] R.N. Wenzel, Ind. Eng. Chem. 28, 988–994 (1936).
[4] A.B.D. Cassie and S. Baxter, Trans. Faraday Soc. 40, 546–551 (1944).



