- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Matter at extremes
- In situ studies of palladium nanoparticles during methane oxidation
In situ studies of palladium nanoparticles during methane oxidation
Uncombusted methane is difficult to remove from the exhausts of natural gas vehicles using catalytic aftertreatment techniques because of methane’s stability, it is the most stable hydrocarbon. A catalyst for the total oxidation of methane, composed of palladium nanoparticles, was studied with the aim of monitoring the composition of the catalytically active material while the reaction occurs.
Methane is the most stable hydrocarbon, its catalytic combustion requires relatively high temperatures to proceed at an acceptable rate. The noble metals palladium and platinum are the most active catalysts for methane oxidation and the metal which performs best generally depends on the operating conditions. For palladium, it has been reported that there are two phases of palladium with high activity for methane oxidation; a reduced phase and an oxidised phase [1]. Here we have investigated methane oxidation over Pd/Al2O3 and Pd/CeO2 catalysts during periodic oxygen pulsing using time-resolved in situ X-ray absorption spectroscopy (XAS) to monitor the composition of the active phase.
X-ray absorption spectra monitoring the Pd K-edge with sub-second time resolution were recorded using the energy dispersive XAS technique at beamline ID24. 2% Pd/Al2O3 and 2% Pd/CeO2 catalysts were subjected to alternating lean and rich feed gas compositions with 0.1% CH4 + 1.5% O2 and 0.1% CH4, respectively. The outlet concentrations of reactants and reaction products were monitored with mass spectrometry.
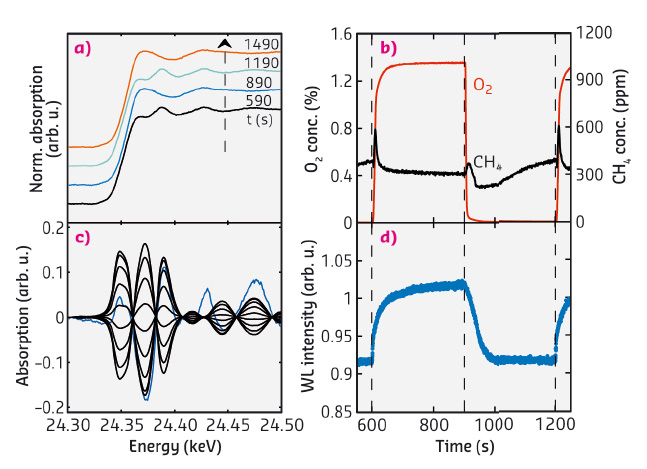
Figure 113 shows results obtained from the Pd/Al2O3 catalyst at 350°C. The near-edge region of the X-ray absorption spectra (Figure 113a) changes reversibly over subsequent rich and lean periods. To analyse the changes in the near edge region, we utilised the phase sensitive detection (PSD) technique [2] which can be used when spectra change reversibly and periodically with time. Using a mathematical transformation, the spectra are transferred from the time domain to the domain of a phase angle and simultaneously static features like the absorption edge and the background are removed. Figure 113c shows the PSD demodulated spectra for phase angles 0-160°. These spectra are similar to the Pd-PdO difference spectrum which shows that oxidation and reduction are the main driving forces behind the changes in the spectra.
 |
|
Fig. 113: Results from oxygen pulse experiments during methane oxidation over Pd/Al2O3 at 350°C. a) XANES spectra, (b) outlet concentration of oxygen (red) and methane (black), (c) demodulated spectra by phase sensitive detection (black) and difference spectrum Pd-PdO (blue), and (d) whiteline intensity at 24.37 keV. |
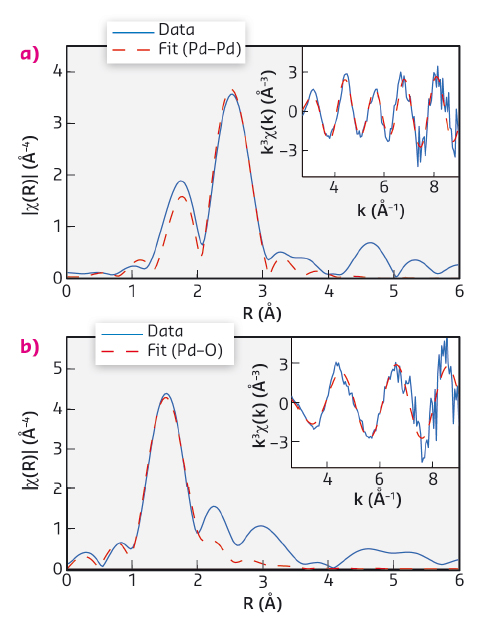
The methane outlet concentration over one pulse sequence is shown in Figure 113b. To follow the oxidation state of the catalyst as a function of time we plot the whiteline intensity in Figure 113d. A first-shell EXAFS fit was performed to further analyse the state of the catalyst at the end of the rich and lean period (Figure 114). The EXAFS fits indicate that at the end of the rich period palladium is in a state similar to metallic Pd while at the end of the lean period palladium is in a state similar to bulk PdO.
 |
|
Fig. 114: (a) Fourier transformed EXAFS spectra and EXAFS fit recorded at: (a) the end of the rich period, and (b) at the end of the lean period. |
At the start of the lean period there is an increase in methane outlet concentration when oxygen builds up on the surface of the reduced Pd catalyst. Palladium is rapidly oxidised, as indicated by the increasing whiteline intensity, also the methane outlet concentration decreases and then holds steady for the remainder of the lean period. At the start of the rich period the methane outlet concentration increases when the oxygen concentration in the gas phase decreases, however, after the reduction of palladium has started, as indicated by the decreasing whiteline intensity, the methane outlet concentration decreases and reaches the lowest value observed during the pulse sequence. In the later parts of the rich period the methane concentration increases when oxygen becomes a limiting reactant.
In conclusion we have used time-resolved in situ X-ray absorption spectroscopy to monitor the state of palladium in supported Pd catalysts during methane oxidation. The highest conversion of methane is observed during the reduction of oxidised palladium to metallic palladium, which indicates that reduced palladium or partially reduced palladium has high activity for methane oxidation.
Principal publication and authors
Chemistry of supported palladium nanoparticles during methane oxidation, J. Nilsson (a,b), P.-A. Carlsson (a,b), S. Fouladvand (a,b), N.M. Martin (a,b), J. Gustafson (c), M.A. Newton (d), E. Lundgren (c) and H. Grönbeck (b,e) and M. Skoglundh (a,b), ACS Catalysis, 5, 2481-2489 (2015); doi: 10.1021/cs502036d.
(a) Department of Chemistry and Chemical Engineering, Chalmers University of Technology, Göteborg (Sweden)
(b) Competence Centre for Catalysis (KCK), Chalmers University of Technology, Göteborg (Sweden)
(c) Division of Synchrotron Radiation Research, Lund University (Sweden)
(d) ESRF
(e) Department of Applied Physics, Chalmers University of Technology, Göteborg (Sweden)
References
[1] A. Hellman et al., J. Phys. Chem. Lett. 3, 678–682 (2012).
[2] D. Ferri et al., Top. Catal. 54, 1070–1078 (2011).



